Primary Antibody, polyclonal or monoclonal antibodies that have been validated for use in Western blots.Cathode Buffer: 25 mM Tris, 20% methanol, 40 mM 6-aminocaproic acid, pH not adjusted.Anode Buffer II: 25 mM Tris, 20% methanol, pH 10.4.Anode Buffer I: 300 mM Tris, 20% methanol, pH 10.4.Gel Running Buffer: 193 mM Glycine, 25 mM Tris (Base), 0.1% SDS.Sample Buffer: 6% SDS, 0.25 M Tris, pH 6.8, 10% glycerol, 10 mM NaF and bromophenol blue with or without 20 mM dithiothreitol (DTT).Filter Paper, cut to same size as gel (Schleicher & Schuell 3MM or equivalent).PVDF Membrane, 0.45 µm pore size cut to same size as gel ( Millipore Catalog # IPVH304F0 or equivalent).Semi-dry Electrophoretic Transfer Cell ( Bio-Rad Catalog #1703940 or equivalent).Please read the following Western blot protocol in its entirety before beginning. Please note that further optimization such as antibody concentration and buffers may be required.  
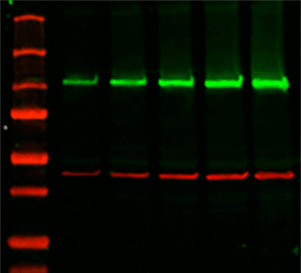
For those proteins that have not been tested with natural samples, a protocol and troubleshooting guide is available for Western blot optimization. The proteins are transferred to a PVDF membrane using a semi-dry transfer apparatus. Protein samples are prepared with SDS and run under both reduced and non-reduced conditions on appropriate SDS-PAGE gel. R&D Systems Quality Control laboratories use this Western blot and immunostaining protocol to show that our antibodies are specific for the protein immunogen. Next, a secondary antibody with an attached enzyme is applied and finally a substrate that reacts with the secondary antibody-bound enzyme is added for detection and visualization. After the proteins are transferred to the membrane, a primary antibody specific for the target of interest is added. Next, the proteins are transferred from the gel to a membrane by electrical current. Western blot works by separating proteins in a sample based on their size using SDS-PAGE gel electrophoresis. Due to this characteristic of antibodies, Western blot analysis can be used to identify and quantify a specific protein in a sample that can contain many different proteins. Antibodies have the ability bind to highly specific sequences in a protein, known as an epitope. The image is captured using a CCD camera.Western blot is a research technique that utilizes antibodies to identify individual proteins within a cell or tissue lysate sample.The membranes are incubated with detection reagent (Immobilon Western Chemiluminescent HRP Substrate, Millipore Corporation, Billerica, MA, USA) for 1 min.To remove residual secondary antibody, the membranes are washed 4 x 5 min in TBST.The secondary antibody (for monoclonal antibodies: HRP-conjugated Goat Anti-Mouse Immunoglobulin for polyclonal antibodies: HRP-conjugated Swine Anti-Rabbit Immunoglobulin, Dako, Glostrup, Denmark) is diluted 1:3000 in blocking buffer and incubated with the membranes for 30 min.To remove residual primary antibody, the membranes are washed 3 x 5 min in TBST (TBS with 0.1% (v/v) Tween20).Optimal dilution must be determined by the user. NOTE: The recommended working dilution of the primary antibody is to be considered as a guideline only. The primary antibody is diluted in blocking buffer and incubated with the blocked membranes for 1 h.To prevent non-specific background binding of the primary and/or secondary antibodies to the membrane, membranes are blocked in a bovine serum albumin-based blocking buffer (2% (w/v) in TBS with 0.1% (v/v) Tween20) for 45 min. Dried membranes from previous steps are activated in methanol for 20 seconds.The proteins are transferred from the gels to PVDF membranes through semi-dry transfer using Trans-Blot® Turbo transfer system (Bio-Rad, Hercules, CA, USA) according to the manufacturer’s protocol.Īll incubation and wash steps are performed at room temperature and with agitation.The electrophoresis is run according to the manufacturer’s protocols. Protein samples are loaded onto Criterion TGX Precast Gels, 4–20% polyacrylamide (Bio-Rad, Hercules, CA, USA).Protein samples (selected tissue lysates, cell lysates or over-expression lysates) are mixed with Laemmli buffer (to a final loading concentration of 2% SDS, 10% glycerol, 0.002% bromophenol blue, 0.0625 M Tris-HCl), supplemented with DTT to a final concentration of 50 mM, and incubated in 95☌ for 5 min. Electrophoresis and blotting Sample preparation Note: This protocol is the recommended Western blot protocol for the Anti-FOS antibody (AMAb91417) as well as the Anti-KLF4 antibodies (AMAb91388 and AMAb91389). Download a pdf version of the protocol for Western Blot - BSA Blocking. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
